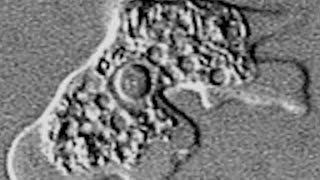
Profounda takes on Leishmaniasis
Profounda licenses Impavido from Knight Therapeutics for marketing into the US. Leishmaniasis is a parasitic disease that is found in parts of the tropics, subtropics, and southern Europe. It is classified as a Neglected Tropical Disease (NTD). Leishmaniasis is caused by infection with Leishmania parasites, which are spread by the bite of phlebotomine sand flies. There are several different forms of leishmaniasis in people. The most common forms are cutaneous leishmaniasis, which causes skin sores, and visceral leishmaniasis, which affects several internal organs (usually spleen, liver, and bone marrow).
The U.S. Food and Drug Administration approved Impavido (miltefosine) to treat a tropical disease called leishmaniasis in March 2014. Leishmaniasis is a disease caused by Leishmania, a parasite which is transmitted to humans through sand fly bites. The disease occurs primarily in people who live in the tropics and subtropics. Most U.S. patients acquire leishmaniasis overseas.
Impavido is an oral medicine approved to treat the three main types of leishmaniasis: visceral leishmaniasis (affects internal organs), cutaneous leishmaniasis (affects the skin) and mucosal leishmaniasis (affects the nose and throat). It is intended for patients 12 years of age and older. Impavido is the first FDA-approved drug to treat cutaneous or mucosal leishmaniasis.
The labeling for Impavido includes a boxed warning to alert patients and health care professionals that the drug can cause fetal harm and therefore should not be given to pregnant women. Health care professionals should advise women to use effective contraception during and for five months after Impavido therapy.
The most common side effects identified in clinical trials were nausea, vomiting, diarrhea, headache, decreased appetite, dizziness, abdominal pain, itching, drowsiness and elevated levels of liver enzymes (transaminases) and creatinine.